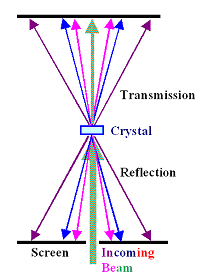
Beams are not monochromatic they are irregular. “Unfortunately, the usual assumptions are unrealistic. “Part of the problem is that many assumptions are made based on an idealized version of a beam,” Wolf continues. “One should measure instead certain correlations collected with averages.” His efforts are reported in Physical Review Letters: “Solution of the Phase Problem in the Theory of Structure Determination of Crystals from X-Ray Diffraction Experiments.” “Instead of trying to measure beam phases - something that is practically impossible and meaningless,” Wolf points out. He shows that by reformulating the problem of phase measurement in diffracted x-ray beams, it might be possible to better study the structures of many materials, providing a more complete picture. Wolf is a researcher at the University of Rochester in Rochester, New York. There is actually data missing to give us a complete picture of many of the structures we are familiar with.” “Even though we have an idea of how DNA and other substances are structured, thanks in part to this process, it is really an approximation. In 1914, shortly after he had left Zurich for Frankfurt am Main, he was awarded the Nobel Prize.“While Laue’s work was very important, it doesn’t provide the complete picture,” Emil Wolf tells. In the two years he spent in Zurich, Max von Laue continued to work successfully on his theory. Max von Laue’s discovery thus laid the foundations for the analytic processes in x-ray crystallography, processes that are today indispensable for research in biology, chemistry, and materials science. The analysis of this specific pattern enabled the researchers to reach precise conclusions about the spatial structure of a crystal. It thus became evident that this was very short-wavelength light.īut not only that: On the basis of von Laue’s discovery, British scientists William und Lawrence Bragg were soon able to demonstrate that the pattern left by the diffracted rays represent a sort of atomic fingerprint of the crystal. They stemmed from the x-rays, which – like visible light through a prism – had been diffracted by the atoms of the crystal. Around the main beam of x-ray light, appearing as large dark spot on the plate, a pattern formed of much smaller spots, some darker, some lighter. Then, in the spring of 1912, what the scientific world had long been seeking became visible on a photographic plate mounted behind the crystal. In a pioneering experiment, von Laue beamed x-rays through a copper-sulfate crystal. With the legendary Munich experiments, von Laue, a mathematician and a physicist, solved in a single stroke two problems that had stymied researchers at the beginning of the 20th century: He clarified the nature of x-rays by showing that, like light, they were electromagnetic waves, and he opened the door for entirely new insights into the atomic structure of matter. Just prior to receiving his appointment in Zurich, this career had gained considerable momentum: In the spring of the same year, in Munich, Laue had conducted the revolutionary experiments that made him famous in the world of physics and, within two years, would bring him the Nobel Prize. Max von Laue arrived Zurich in October 1912 to commence the first professorship of his academic career. Max von Laue solved two longstanding problems with a single experiment: He clarified the nature of x-rays and laid the cornerstone for a procedure that reveals the atomic structure of matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
